Review Interventional
Percutaneous
transhepatic image-guided interventions for malignant biliary obstructions:
Current status
Adam
Hatzidakis1, Miltiadis Krokidis2
1Department of Medical Imaging - Heraklion University
Hospital, Medical School-University of Crete, Heraklion, Greece
2Department of Radiology, Cambridge University
Hospitals, Cambridge, United Kingdom
Submission: 18/1/2017 | Acceptance: 4/5/2017
Abstract
A malignant biliary
obstruction (MBO) occurs when there is a blockage of the bile outflow towards
the duodenum due to a malignant tumour. The most common tumours that cause
MBO with direct invasion of the biliary tree are pancreatic carcinoma and
cholangiocarcinoma. The biliary tree may also be blocked from tumours that
cause external compression, such as enlarged hilar or ampulary lymph nodes or
in some cases of hepatocellular, gastric or gallbladder cancer. Surgery is the
treatment of choice if the disease is at an early stage and adjacent structures
are not infiltrated. Otherwise patients will receive palliative treatment for
quality of life improvement. Percutaneous transhepatic image-guided biliary
interventions offer a minimal invasive approach that decompresses the blocked
biliary system and have an established role in the management of both operable
and inoperable patients with MBO.
A variety of devices and techniques have
been developed for this purpose, including the use of internal and external
drains, plastic, bare metallic and covered metallic stents, biopsy forceps and
unilateral or bilateral, one- or two stage- approach. The purpose of this
review article is to offer a global overview of the interventional radiology
role in such patients and to discuss the latest developments.
Key
words
Percutaneous interventions; Biliary
malignancies; Biliary drainage; Metallic stenting; Interventional radiology
Corresponding Author,
Guarantor
Adam Hatzidakis MD, Ph.D, EBIR
Associate Professor of Vascular and
Interventional Radiology
Department
of Medical Imaging, Heraklion University Hospital, 71500, Heraklion, Greece
E-mail:
adamhatz@hotmail.com
1. Introduction
The tumours that may lead to malignant
biliary obstructions (MBOs) are mainly adenocarcinoma of the pancreatic head
and extrahepatic cholangiocarcinoma [1]. Other tumours that may lead to
malignant jaundice are ampulary, gallbladder, and hepatocellular carcinoma and
lymph node strictures from gastric, pulmonary, breast and oesophageal cancer.
When malignant jaundice occurs, the normal bile flow towards the duodenum is
blocked. Although the complete cholestasis mechanism is not fully interpreted,
the lack of bile in the duodenum results in a systemic inflammatory reaction
due to release of cytokines that targets specific organs hence leading to
multi-organ failure. The deranged liver function leads to Kupffer cells
dysfunction and the lack of bile in bowel increases the membrane permeability
and reduces the bowel bacterial barrier, so promoting bacterial migration,
initially to the portal and then to the systemic circulation [2-4]. If this
situation is not corrected, uncontrollable sepsis occurs very quickly in the
majority of cases.
2. Management of patients with MBOs
Patients with a MBO would either be
considered operable or inoperable according to the stage of disease at diagnosis.
Endoscopic drainage is usually the first approach in most of the centres.
However this is not always technically feasible, particularly for lesions
located proximally to the liver hilum [5]. Furthermore, when preoperative
drainage is required there is an increased risk of infectious complications
with the endoscopic approach [6]. The main contraindications for endoscopic
retrograde cholangiopancreatography (ERCP) are the presence of an oesophageal
stricture, gastric volvulus, bowel perforation, unstable cardiopulmonary
status that would prohibit patient to be positioned supine and history of
previous bowel surgery with change of the anatomy (Billroth II or Roux-en-Y
loop). Therefore percutaneous transhepatic drainage access should be
considered as the most appropriate treatment option [7]. Once the biliary tree
is decompressed and biopsy of the underlying lesion is obtained, a multidisciplinary
team decides on whether to proceed with surgical resection or palliative
treatment. In the case of palliative approach, internalisation of the drains
with the use of stents is required in order to reduce infection risk during
chemotherapy. Stents ideally need to be patent for the whole patient’s life
span, in order to avoid treatment interruption or re-intervention for
cholangitis. The management algorithm of a MBO patient is shown in Table 1.
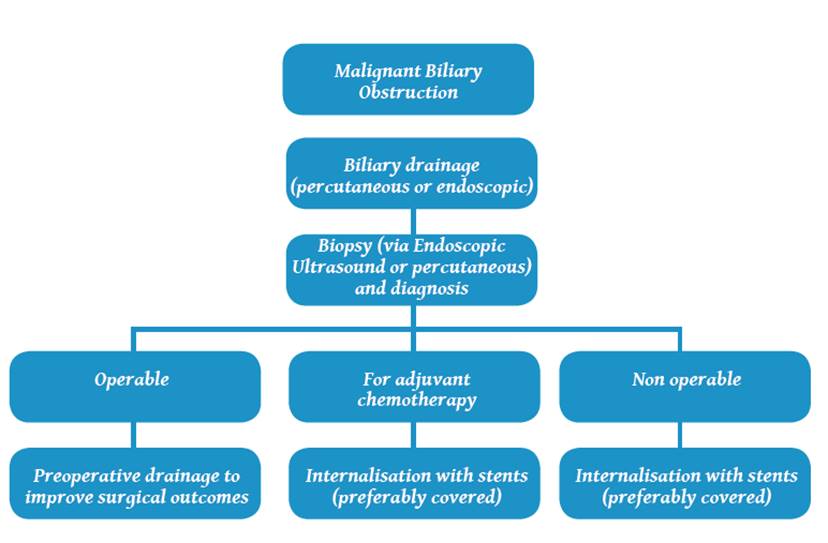
Table
1. Suggested
management algorithm of patients with malignant biliary obstructions
3. Diagnostic Imaging
Initial imaging approach of MBOs is
performed with trans abdominal ultrasound (US) that is expected to detect the
dilated bile ducts and the presence of possible intrahepatic deposits. US is
quick, accessible and of low cost. However some pitfalls may occur in the case
of obese patients, when there is bowel interposition or in the case of
paralysis of the right hemidiaphragm.
Computed tomography (CT) is usually
performed not only to delineate the stricture but also to assess the presence
of intrahepatic and distal metastatic deposits. A triple phase CT scan is
usually recommended for characterisation of the malignant stricture, even
though malignant biliary tumours are not expected to be enhancing avidly in
the arterial phase. Adding a late contrast phase (6-15 min post injection)
appears to increase the sensitivity of detection of adenocarcinomas [8].
Magnetic resonance imaging (MRI) plays
also an important role in the diagnosis of malignant biliary strictures,
particularly if combined with magnetic resonance cholangiopancreatography
(MRCP); the latter is a T2-weighted fast-spin echo sequence applied with prolonged
effective echo time (>240 milliseconds). The signal from static fluid
(biliary tree) is enhanced and the signal from the surroundings structures is
suppressed (Fig. 1a). MRCP is able to locate the level of obstruction in
85-100% of the cases. In addition, with the use of conventional MRI sequences
the sensitivity and the specificity for type of tumour diagnosis reach 86% and
98% respectively [9].
Malignant strictures involving the hilum
are classified using the Bismuth-Corlette classification system based on the
extension of the stricture into the intrahepatic ducts [10]. Bismuth type I
strictures involve the proximal common hepatic duct and spare the confluence
between the left and right ductal systems; type II strictures involve the
confluence and spare the segmental hepatic ducts; types IIIa and IIIb involve
either the right or left segmental hepatic ducts, respectively; and type IV
strictures involve the confluence and both the right and left segmental
hepatic ducts.
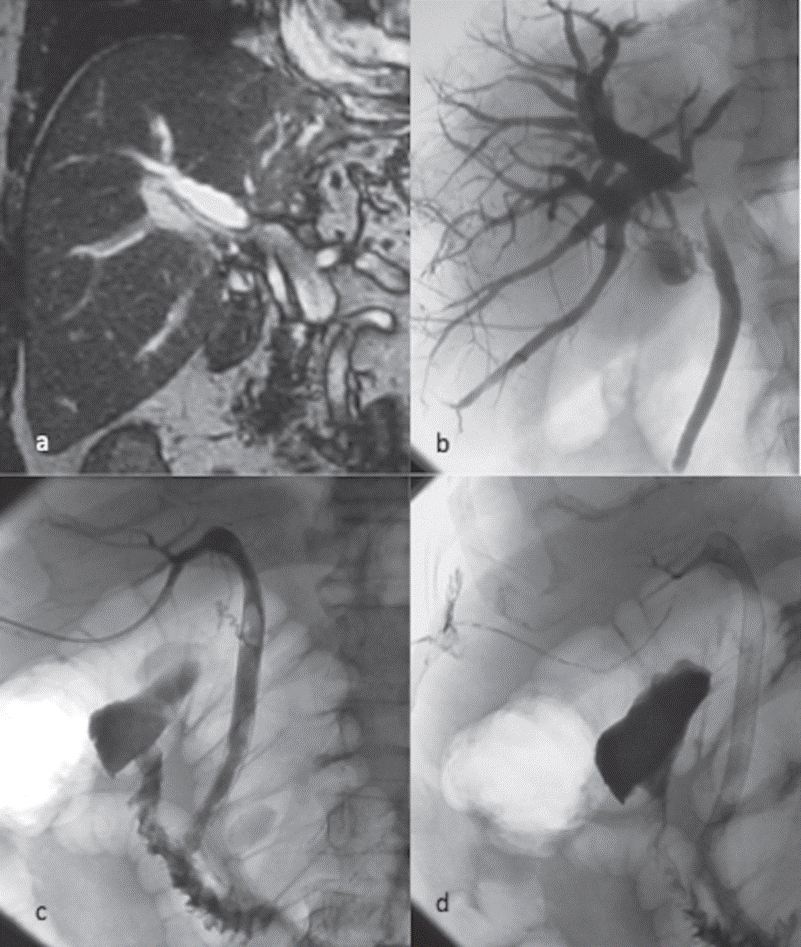
Fig. 1.
(a) Coronal MRI of the upper abdomen reveals marked intrahepatic biliary
system dilatation and an occlusion at the liver hilum.
(b) A hilar lesion is confirmed after transhepatic cholangiography. Distal
common bile duct and papilla are free from tumour infiltration.
(c) Palliative treatment is completed with the primary placement of a
metallic stent, which preserves the obstructed biliary duct patent, without
need for a biliary catheter. The sphincter of Oddi is not stented in this case.
Cholangiographic control via 5Fr catheter that is left in situ is performed.
(d) Transhepatic embolisation can be performed
by injection of n-butyl cyanoacrylate mixed with Lipiodol along the tract
4. Percutaneous approach and drainage
The main indication for percutaneous
transhepatic biliary drainage in a patient with a malignant biliary stricture
is the elevation of bilirubin from a mechanical cause. The procedure may be
performed on an emergency basis if jaundice is combined with cholangitis and
sepsis. Contraindications are mainly technical and are usually relative like
i.e. the coagulation status of the patient or the presence or not of ascites,
with the exemption of complete infiltration of the liver parenchyma from widespread
metastatic disease (Table 2).

Assessment of the coagulation status of
the patient is of paramount importance prior to the procedure. In case of
deranged clotting, blood products (vitamin k, fresh frozen plasma or platelet
transfusion) may be administered. In case of presence of ascites, percutaneous
drainage may be performed prior to accessing of the biliary tree. The procedure
is usually performed under local anaesthesia (lidocaine 2%) and conscious
sedation using 1-8 mg of midazolam and 50-200 μg of fentanyl. Antibiotic prophylaxis
(i.e. with cefuroxime 750 mg) may be administered before the procedure and
continued for up to 5 days after the procedure according to the operator’s
preference.
Percutaneous approach is performed by a
transhepatic puncture with a rather thin (21-22 G) Chiba needle under US or
fluoroscopic guidance. When access to a bile duct is obtained a minor amount of
diluted contrast is usually injected to confirm correct position. Okuda et
al. first described the fluoroscopically guided technique, in 1974 [11]. When
the thin needle was positioned in the biliary system a cholangiogram was
performed and a second puncture followed to a duct that was considered
adequate in terms of angulation and size. The second puncture was performed
with a 5 Fr needle catheter and a 0.035” inch wire was advanced in the biliary
tree. With the use of US guidance, once the thin needle is in the biliary
system a 0.021” inch wire is advanced in the biliary tree. The system is
upsized to 6 Fr without the need of a second puncture in this case. When the 6
Fr catheter is advanced a cholangiography is performed with diluted iodinated
contrast (Fig. 2a) [4]. For common bile duct (CBD) lesions, right-sided
approach is preferred, except in cases of ascites or colon interposition (Fig.
1b). Right side route provides a more straight way for wire and catheter
manipulation and keeps operator’s hands away from x-ray beam (Fig. 2b).
Left sideapproach on the other hand appears less painful for the patient as
there is no route through intercostal spaces. For hilar lesions the puncture
site should be decided after studying tumour location, extent and liver lobe
infiltration, potential lobe atrophy and biliary tree anatomical variability
[12].
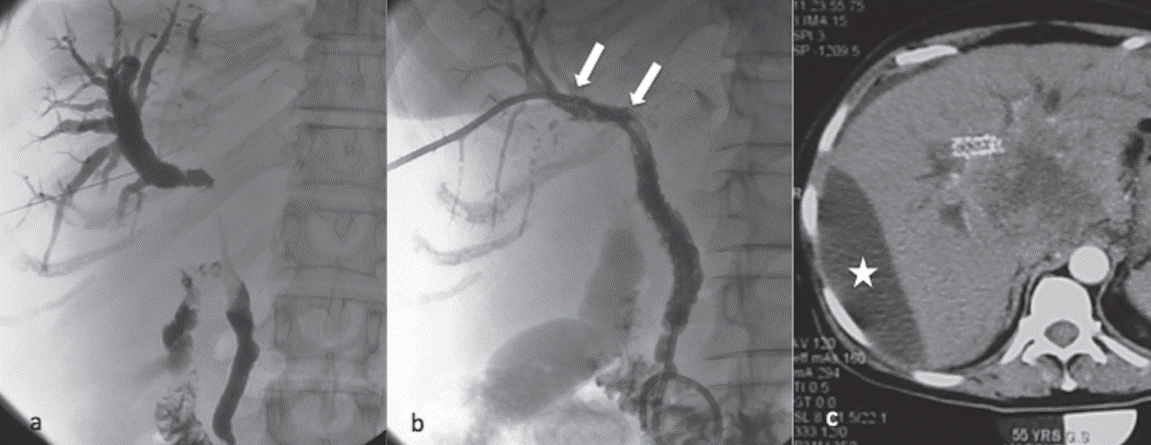
Fig. 2. (a) Transhepatic puncture with a thin 21 G Chiba needle, aiming for the
intrahepatic biliary tract system. After filling with diluted contrast medium,
the malignant stenosis is revealed. Small amount of contrast crosses the
stricture and opacifies the distal CBD and the duodenum. (b) The same
patient after metallic stent placement. No stricture is seen due to the fully
expanded metallic stents. Two covered stents were required to preserve the long
stricture open. Notice the two upper radiopaque markers in the proximal stent.
Between the two markers, stent’s coverage has side holes for maintaining
intrahepatic bile ducts patent. (c) Large subcapsular biloma is seen in
follow-up CT (white star). Bile leakage occurred through the parenchymal tract
after catheter removal
In case of opacification of multiple
obstructed bile ducts, the operator should try to drain as many of the
opacified ducts as possible in order to avoid bacterial contamination and post
procedural contrast related cholangitis [13].
In the case of MBOs, percutaneous transhepatic
biliary drainage (PTBD) follows in order to decompress the obstructed bile
duct system [14]. The drainage of the bile ducts is usually performed with a
small 8 Fr plastic multi-hole pigtail catheter. In the cases where the lesion
has not been biopsied or in the case where the biliary tree is infected,
external drainage catheter placement is suggested. The catheter is secured to
the skin with sutures. Self-locking catheters are preferred in order to
minimise the dislocation risk. In cases of complex hilar strictures, placement
of multiple external biliary catheters may be necessary to achieve complete
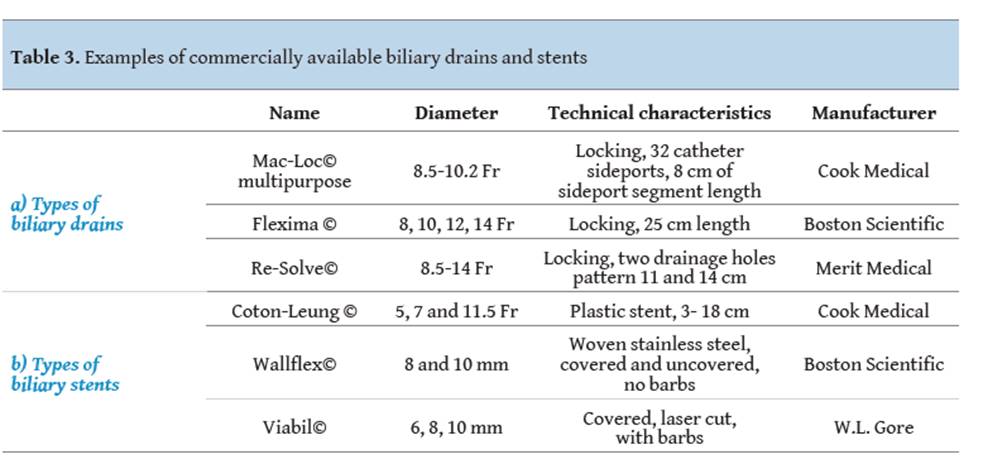
drainage. The types of available drains are described in Table 3a.
Biopsy of the lesion may be either
obtained with endoscopic ultrasound - guided fine-needle aspiration (EUS-FNA)
or with the use of endobiliary forceps. EUS-FNA is effective in obtaining
cytological samples for Bismuth I-II lesions, however for hilar lesions this
access is less effective [15, 16]. In addition, the cytologic sample may not
always be diagnostic and core biopsy of the lesion may be required. Endobiliary
biopsy may be obtained with theuse of biopsy forceps overcoming the problem of
inadequate sampling that is encountered with FNA [17]. The forceps may be
advanced either endoscopically or percutaneously. In the case of percutaneous
insertion a 7Fr sheath is used as access and a security wire is used and placed
across the stricture. The forceps are placed on the side of the wire and
usually 3-4 samples are obtained and placed in a formalin suspension.
If there is
already confirmation of malignancy and there is intention to proceed with a
stent the obstruction can be crossed with a hydrophilic wire and the percutaneous
biliary catheter can be pushed through the stenosed /obstructed duct, so that
bile is draining both internally towards the duodenum and externally (internal-
external drainage) (Fig. 3a) [4, 5]. By crossing the stricture a “rendezvous”
procedure with endoscopic approach may also be performed particularly for
Bismuth III and IV lesions.
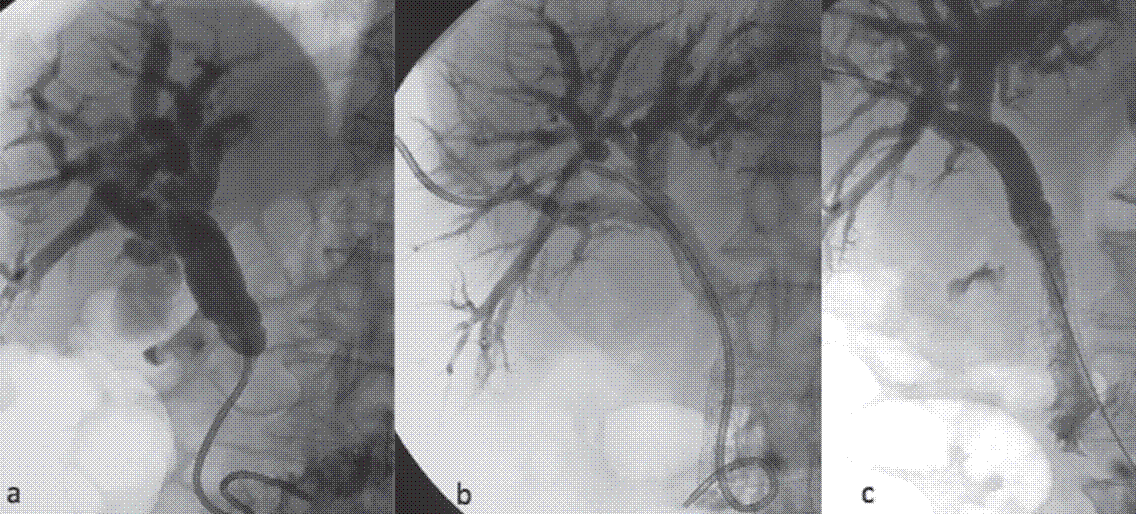
Fig. 3. (a) A hydrophilic wire has crossed the distal CBD
obstruction and an 8 Fr biliary catheter was advanced with the distal tip in
the duodenum.
(b) Two weeks after stent placement, cholangiography reveals stent
occlusion.
(c) After
“cleaning” with an angioplasty balloon it is assumed that occlusion was due to
bile sludge incrustation
5. Use of biliary stents
In case of inoperable tumours,
internalisation of the drain is required in order to be able to either
administer palliative chemotherapy or to offer a satisfactory quality of life
[18-20]. This is achieved with the use of stents that may be plastic, bare metallic
or covered metallic (Table 3b). Prognostic indicators for stent
placement are thegeneral status of the patient, the performance score of
oncologic measures (i.e. the Karnofsky performance status scale) and the
status of the organ as indicated by the liver biochemistry.
Metallic biliary stents have been proved
as the best palliative treatment of non-resectable malignant obstructive
jaundice, allowing longer patency rates than plastic endoprostheses [21]. The
technique is safe, with a high technical success rate of over 97% and low complication
rate [22, 23] and procedure related mortality is reported between 0.8 and 3.4%.
Early complications rate within the first 30 days is about 2% while the late
complications rate can reach 16% [23]. Mean overall primary stent patency is
reported 120 days, but mean overall secondary stent patency is 242.2 days [22].
An integration process accompanies the
deployment of metal endoprostheses within the biliary tree into the surrounding
malignancy, which after a period leads to the formation of tissue through the
stent’s struts that finally blocks the stent. This is the so-called “tumour ingrowth”.
Ingrowth is the result of tumour growth but also of the accumulation of biliary
sludge and bacterial biofilm. The motility of bare stents plays a crucial role
on the latter aspect of ingrowth as “fractures” of the stents occur and lead to
dysfunction. This effect occurs more frequently with laser cut metallic stents
therefore the use of woven stents is suggested.
The malignant proliferation can sometimes
occur in the proximal end of the stent and not through the mesh. This is called
“tumour overgrowth” and also gradually blocks the stent lumen [24]. In such
cases, patient requires new percutaneous intervention that leads to placement
of a second stent through the occluded stent. In some cases stent occlusion is
due to bile sludge and needs “cleaning” with a simple angioplasty balloon (Fig.
3b-c).
Hausegger et al. [25] in the early days of
use of metallic stents in the biliary system analysed the histological changes
after the deployment of stainless steel endoprostheses in the biliary tree for
the treatment of malignant biliary disease. In fourteen cases histological
examination was performed after autopsy and in one case a surgical specimen
was analysed after tumour resection. In the analysed specimens, the stent was
inserted in a period ranging from 5 days to 21 months. For the initial period
of the first 15 days, histology revealed that the cuboid epithelium of the
biliary tree is completely destroyed in the areas that were in contact with the
stent. There were moderate inflammatory changes in the sub mucosa with minor
lymphatic infiltration and oedema. The internal layer of the stent was covered
by non-specificgranulomatous tissue. The fibrous tissue and the tumour were
displaced. No tumour cells were recognised within the biliary tree lumen and no
signs of acute inflammation were noticed. In the period between the 2nd and
the 12th month the endoprosthesis was gradually integrated to the surrounding
tissue by a layer of granulomatous tissue and tumour ingrowth. In a similar
study Boguth et al. [26] describe similar histological changes that occur in
the first 3 to 6 months and lead to the occlusion of the bare metallic
endoprosthesis. Ingrowth through the mesh of the bare stent occurs in all
patients that survive more than 6 months and re-intervention is usually required.
The patients may present with cholangitis and a new procedure and a new stent
placement is usually necessary to resolve the situation.
There is usually a difference in timing of
clinical expression of symptoms that is related to the location of the tumour.
Intrahepatic lesions tend to give symptoms later than extrahepatic ones. This
is due to the fact that the only symptoms expected are those related to the malignant
obstruction of the biliary tree and in case of intrahepatic tumour, there are
several collateral drains that may be used until complete occlusion occurs
[27].
6. Covered stents
In order to reduce stent’s dysfunction
from tumour ingrowth, covered metallic stents were developed in the last
decade. Various authors tested several coverage materials with a different
range of results [27]. The initially used covered stents were “home made” by
applying a coverage membrane on the available bare stents. Saito et al. in
1994 used biliary Gianturco-Roesch Z-stents covered with a Gore-Tex membrane
[28] and reported satisfactory medium- to long-term results in a study of six
patients. Thurnher at al. reported in 1996 their experience with the first
type of covered Wallstents [29]. The coverage was a 0.015 mm thick polyurethane
membrane that was also used from Rossi et al. in 1997 [30] and Hausseger et
al. in 1998 [31]. Both investigator groups reported that the 0.015 mm
polyurethane membrane was eroded from tumour and gastric juice. Similar results
were also presented from Kanasaki et al. in 2000, where nitinol Strecker stents
were used with the same coverage [32]. A 0.035 mm polyurethane membrane was
used in homemade covered Gianturco-Roesch Z-stents and spiral Z-stents from
Miyayama et al. in 1997, with better results [33]. Han et al. reported a 71%
patency at 20 weeks using a 0.030 mm thick polyurethane membrane in covered
Niti-S stents [34]. Isayama et al., using 0.040-0.050 mm thick
polyurethane-covered Wallstents, did not report tumor ingrowth [35] and
presented even improved results with a 0.050-0.060 mm polyurethane membrane
using covered Diamond stents [36].
During the last ten years, covered stents
with a coverage membrane from expanded poly-tetra-fluoro-ethylene/
fluorinated-ethylene-propylene (ePTFE/FEP) were developed and are available in
the market (Viabil©, W. L. Gore & Associates, Flagstaff, AZ, USA) [24].
Randomised controlled trials were performed with the use of such covered
devices that have shown the superiority of these covered stents in comparison
to uncovered stents in specific patient population (Fig. 2a-b) [37,
38].
Covered stents are not suitable for all patients
with malignant jaundice. They have to be reserved for patients with a
reasonable expected survival and not used in the case of advanced disease [39].
Furthermore, anatomical features have to be taken into consideration including
the stricture site, location and patency of the intrahepatic, cystic and
pancreatic ducts. Usually, only Bismuth type I strictures are suitable for
covered stent placement, whereas specific covered stents may be placed in some
cases of type II. The covered portion should not be advanced in the
intrahepatic ducts in order to avoid cholangitis. For this purpose, covered
stents with side holes have been developed. The “holed” region does not prevent
tumour ingrowth and it is also not extending proximally enough to prevent
tumour overgrowth as a bare stent would. Nevertheless, side holes permit
placement in anatomically complicated cases avoiding cholangitis or
cholecystitis. The same principal should be followed for the cystic duct but
less for the pancreatic, since pancreatitis may less frequently occur and the
location of pancreatic duct is not a true limit in stent placement.
7. Stent placement techniques
Stent placement can be performed as one-
(so called “primary stenting”) or two- (or more) step procedure. The factors
that would influence the type approach are multiple but mainly consist of the
presence or not of diagnosis of malignancy, the presence or not of biliary
sepsis and technical issues such as intra-procedural bleeding or bile leak.
If a more than one step approach is
decided then a biliary drainage catheter may be left in situ for 1-2
weeksbefore a stent is inserted. Adam et al. [40] introduced the concept of
“primary stenting” in 1991 (Fig. 1b-c). They suggested that
self-expandable metallic stents could be placed in patients with a better
prognosis as a one-step procedure, aiming to reduce total hospitalisation time.
However, at the time operators were inclined to check with a cholangiogram 1-3
days later. Therefore they suggested leaving in situ a 5 Fr angiographic catheter
as access for the cholangiographic control (Fig. 1c). The catheter was
retrieved, after stent’s expansion was confirmed and complications were
excluded (Figure 4c-d). This is a practice that many operators still follow
nowadays, even though true “primary stenting” consists of obtaining access,
crossing the lesion, deploying a stent and plugging the tract in one session (Fig.
1d). To achieve that, diagnosis needs to be obtained prior to the biliary
procedure i.e. previous EUS-FNA and the whole biliary procedure needs to be
uneventful without bleeding or biliary leak. Intra-procedural remodelling of
the stent is required to ensure that expansion is satisfactory. If so the tract
can be plugged with gelfoam pellets and the patient can be discharged the next
day. The whole procedure may last 1-2 hours, depending on the degree of
anatomical complexity. The procedure may be performed under deep sedation and
analgesia or general anaesthesia [41]. Hospitalisation time depends on the complexity
of the case and the manoeuvres performed. In general, patient is kept in
hospital until he/she fully recovers. Immediate post-procedure monitoring
consists of haemodynamic assessment and puncture site review.
In the past years, there was discussion
about the usefulness of bilateral versus unilateral lobe drainage and
stenting. In case of bilateral drainage, stents may either be positioned by
puncturing separately the right and left side ducts and catheterising
separately the CBD (Y configuration), or may be placed from a single side
puncture after catheterising the other side ducts and the CBD form the same
side (T configuration). There is still a degree of controversy as to whether
partial or complete biliary drainage should be done. Inal et al. [42] studied
138 patients with hilar malignant strictures that received unilateral or
bilateral stenting. Only patients with type IV lesions appeared to benefit from
bilateral stenting, whereas for those with Bismuth type II and III there was
no benefit in terms of patency. Although the cumulative stent patency seemed to
be better after bilateral than unilateral drainage approach, there is, based
on the available literature, not enough data to support bilateral drainage for
malignant hilar obstruction [43]. Older and recent studies show that
partial-liver drainage achieves results as good as those after complete liver
drainage with significant improvements in quality of life and reduction of the
bilirubin level [22, 44, 45]. Therefore the insertion of more than one stent
would not appear justified as a routine procedure in patients with biliary
bifurcation tumours.
Another frequently asked question was if
we should stent the sphincter of Oddi in every case, even if the tumour is
ending higher than the level of the papilla. A study performed in 2001 showed
that in patients with extrahepatic lesions lying higher than 2 cm from the
papilla and with a relative poor prognosis (<3 months), due to more advanced
disease or to a worse general condition, the sphincter of Oddi should be also
stented in order to reduce the post-procedural morbidity (Fig. 1b-c)
[20].
8. Plastic vs. metallic stents
Bare self-expandable metallic stents have
a mean patency of approximately 6-8 months and this is superior to what the
plastic endoprostheses offer (Fig. 1c) [21, 46]. According to Lammer et
al. [21], the use of self-expanding metal stents appears to show substantial
benefits for patients with MBOs and also appears to be more cost-effective
than the use of plastic ones, even though the individual plastic stent is
significantly cheaper than the metallic one. The cost-effectiveness lays on the
fact that the patency is higher and the number of repeated interventions
significantly lower [46]. A recently published meta-analysis includes results
from 785 patients in 10 randomised trials half of which received a metallic
stent and half a plastic one [47]. The results showed that metal stents were
associated with a significantly longer patency, smaller number of re-interventions
and longer survival of the patient, indicating once again that plastic stents
do not offer any significant benefit for MBOs.
9. Bare vs. Covered stents
What is of interest is the comparison
between uncovered and covered metallic stents. ePTFE/FEP covered stents were
used in two randomised trials published in the literature. In the first trial
[38], covered stents were directly compared with uncovered in patients with
Bismuth type I cholangiocarcinoma. Sixty patients (36 men and 24 women, with age
range 46-78 years) were randomised with the use of a sealed envelope for the
placement of a covered or a bare stent. In 21 cases the tumour also infiltrated
the cystic duct. Patients were followed-up with telephone interviews and on an
outpatient basis. Technical success was 100% for both groups. Minor early
complications were noticed in 13.3% of the bare stent group and 10% of the
patients of the covered stent group. The mean follow-up period was 212 days
(45-675 days) and all patients had passed away at the end of the study.
Thirty-day mortality was zero for both groups. Median survival time was 180.5
days for the bare stents and 243.5 days for the covered stents, with p<0.05.
Stent’s mean patency rate was 166 days for the mesh stent and 227.3 days for
the covered stent, with p<0.05. Stent dysfunction occurred in 9
patients with bare stent after a mean period of 133.1 days and forceps biopsy
revealed ingrowth in 88.8%. Dysfunction occurred also in 4 cases of the
covered stent group after a mean period of 179.5 days and it was due to tumour
overgrowth in 2 and due to sludge incrustation in another 2. Tumour ingrowth
occurred exclusively in the mesh stent group. There was also no difference in
the overall cost of the two groups after a cost analysis.
The second prospective randomised trial
that compared ePTFE/FEP covered stents with bare stents was performed in
patients with pancreatic adenocarcinoma and revealed similar results [37]. The
study was performed in 80 patients that were also randomised with the use of a
sealed envelope into a bare stent and a covered stent group. The patients were
53 men and 27 women with an age range from 41 to 79 years (mean 62.7 years).
Technical success was 100% in both groups. Early complications were observed
in 10% of the bare and 12.5% of the covered stent group. Median follow-up time
was 192 days (range of 104-603 days); all patients passed away by the end of
the study. The 30-day mortality was zero for both groups. Median survival time
was 203.2 days for the bare stent group and 247 days for the covered stent
group, and this difference was not statistically significant. Mean primary
patency was 166 days for the uncovered and 234 days for the covered stents,
with p< 0.05. Dysfunction occurred in 12 bare stents after a mean period of
82.9 days and it was due to tumour ingrowth in 91.6% of the cases. Dysfunction
occurred in 4 covered stents after a mean period of 126.5 days and it was due
to tumour overgrowth in 2 and due to sludge in 2. Cost analysis revealed that
there was no difference in the overall cost of the two groups. The two
randomised trials showed that the ePTFE/FEP covered metal stents appear to
reduce significantly the rate of stent’s dysfunction. The micro porous membrane
appears to limit completely the risk of tumour ingrowth, which is the main
issue of the use of metallic endoprosthesis in malignant biliary obstruction.
However, in order to benefit from the results of covered stents patient’s
survival needs to be long enough for ingrowth to occur. If survival of more
than six months can be predicted -by the lack of metastatic disease and the
performance status of the patients- then use of an ePTFE/FEP-covered stent is
completely justified. The mentioned device appears to limit also another of
the major problems of the covered stents which is stents’ migration, by having
the lateral barbs (anchoring fins).
There are still some investigations going
on about prevention of “overgrowth”. An improvement might be achieved if
covered stent placement could be combined with bare stent extension proximal
and distal to the coverage. In this manner, the papilla could be stented with
bare metal, so that potential episode of acute pancreatitis is avoided and also
tumour overgrowth can be delayed due to the bare stent above the tumour area (Fig.
4a-d).
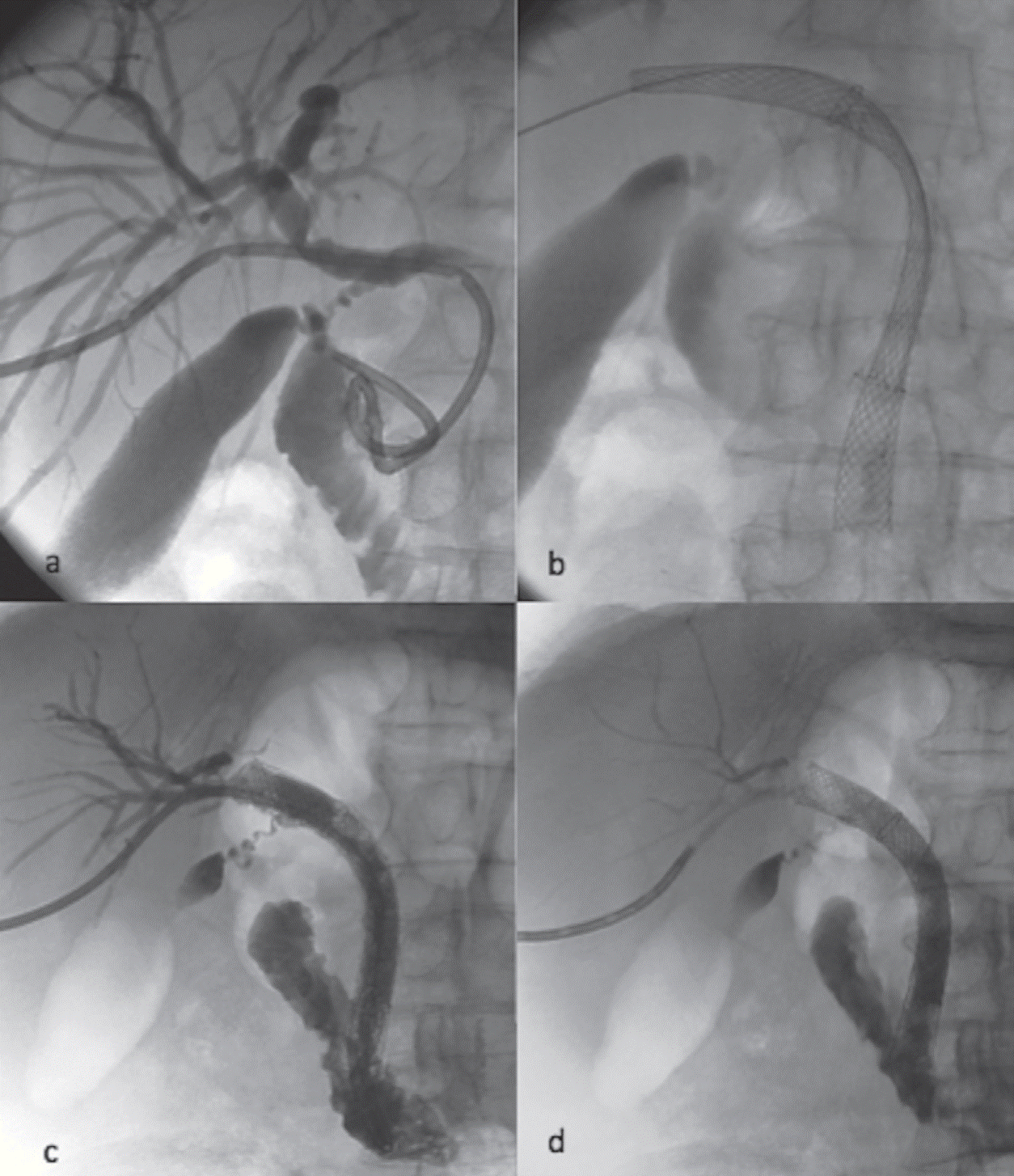
Fig. 4.
(a) Internal drainage with an 8 Fr biliary catheter. Malignant occlusion is
in the distal CBD area above pancreatic head, due to a cholangiocarcinoma.
(b) After covered stent placement at the level of the obstruction, a second
long uncovered metallic stent was placed inside the covered one.
(c) In this manner, the papilla could be stented with bare metal, so that
potential episode of acute pancreatitis is avoided and also tumour overgrowth
can be delayed due to the bare stent above the tumour area.
(d) The catheter can be retrieved after we are
confident that the stent is fully expanded and that no other major
complications like arterial bleeding, sepsis or stent malfunction occur
10. Percutaneous vs. Endoscopic drainage
The decision on whether to obtain an
endoscopic or a percutaneous access to a blocked biliary system has been based
mainly on local expertise and availability. There is very limited comparison of
the two approaches in the literature. The area where the two methods have been
more extensively compared is the preoperative biliary drainage where the
endoscopic approach is considered to jeopardise the aseptic biliary environment
and lead to infections of patients that will be operated. The existing studies
have been analysed in a very recently published meta- analysis with a focus on
patients with resectable perihilar cholangiocarcinoma [48]. The authors
included four retrospective studies on 433 patients; 275 (63.5%) underwent an
endoscopic drainage and 158 (36.5%) a percutaneous. The percutaneous approach
has demonstrated to offer superior results in terms of procedure-related
morbidity and rate of conversion from one procedure to the other and
cholangitis rate whereas pancreatitis occurred exclusively in the endoscopic
group.
In a very recent publication the results
of EUS are compared with the use of percutaneous transhepatic cholangiographic
endobiliary forceps biopsy (PTC-EFB) in termsof diagnostic samples performance
in 137 patients in a retrospective cohort study showing similar sensitivity,
negative predictive value and accuracy [49]. The authors concluded that
endobiliary biopsy should be the treatment of choice in case biliary drainage
is also required.
11. Complications and how to deal with
them
The most common minor complications are
pain, stent migration, stent insufficient expansion and fever. Pain is treated
or prevented by IV or IM administration of analgesics and/or sedatives [41].
Stent insufficient expansion can be corrected by post-stenting balloon dilatation.
Stent misplacement or migration can be corrected by placement of a second
stent.
Most feared major complications are
sepsis, bleeding and bile leakage. As mentioned above, complication rate within
the first 30 days is about 2%, while late complications rate can reach 16%
[23]. Comparing uncovered to covered stents, minor early complications were
noticed in 10-13.3% of the bare stent group and 10-12.5% of the patients of the
covered stent group, with a 30-day mortality of 0% for both groups [37, 38].
In order to prevent such serious
complications, biliary interventions should be performed under IV antibiotic
coverage [50]. Any biliary intervention is considered at the minimum a
clean-contaminated procedure and therefore the recommendation is that all
patients scheduled for biliary drainage receive prophylactic antibiotics prior
to the procedure [50-52]. Transient bacteraemia occurs in approximately 2% of
patients after biliary intervention [51]. If a patient develops fever and/or
chills following biliary intervention, antibiotics may be continued, fluid
resuscitation should be initiated and the need for blood cultures considered.
In some cases, infection does not respond to these measures and additional
drainage may be required to address incompletely drained or isolated bile
segments. In patients with sub-segmental isolation, multiple drains could
potentially be required and long-term antibiotic suppression may be favoured
[52].
Arterial bleeding is a relatively rare complication
of PTBD, appearing in 0.6-2.3% and when it does not resolve spontaneously, it
should be treated by selective arterial embolisation [53-55]. Arterial
complications might be prevented by obtaining access from second or third order
ducts, located in the periphery of the liver and not near the hilum. Central
punctures might be complicated with portal vein and/or arterial injury that
will be manifested with haemobilia and/or pseudoaneurysm formation. It
appears that there is a higher incidence of haemobilia associated with left
lobe puncture, but did not reach the threshold of statistical significance (p=0.077)
in previous studies [56, 57]. An emergency angiography should be considered in
all patients in whom a pseudoaneurysm is suspected following hepatobiliary
interventions. Transcatheter arterial coil embolisation is a safe and effective
treatment for pseudoaneurysm with a technical success rate of 95.8% [57, 58].
Minor complications can be observed after embolisation in 80.6% patients, 76.4%
of whom may have hepatic ischaemia and 4.2% focal hepatic infarction [57].
Surgical intervention should be reserved for patients for whom embolisation is
not possible or fails [59].
Bile leakage can occur through the
parenchymal tract after catheter removal (Fig. 2b-c). In order to
prevent this, several embolisation techniques have been proposed [60-62]. One
is by placing small pieces of expandable gelatin foam pellet (Hunter Biopsy
Sealing Device, Vascular Solutions, MO, USA) [59]. The authors report that utilisation
of this method has the potential to reduce the morbidity and mortality burden
associated with post-PTC haemorrhage by preventing bleeding from the liver access
tract. The same kind of embolisation can be achieved by injection of n-butyl
cyanoacrylate mixed with Lipiodol (Fig. 1c-d) [62]. A relatively newer
device is Hep-Plug sealing (IFU, Vascular Solutions, MO, USA), which works in a
similar way as the Hunter Biopsy Sealing Device.
12. Conclusions and future perspectives
Percutaneous transhepatic biliary
procedures are integrated in the management of patients with MBOs. In case of
operable disease preoperative biliary drainage may be performed - with better
results than the endoscopic approach- offering decompression of the biliary
tree and access for endobiliary biopsy. In case of palliative approach either
woven bare stents or covered stents may be used to alleviate jaundice for the
patient’s life span. Such procedures have to be part of the everyday
armamentarium of interventional radiology centres. Future perspectives will
probably be in the direction of smaller profile and functional (or “drug
eluting”) stents and endobiliary ablation treatment that are still in a very
early stage. R
Conflict of interest:
The authors declared no conflicts of
interest.
References
1. Wade
TP, Prasad CN, Virgo KS, et al. Experience with distal bile duct cancers in
U.S. Veterans Affairs hospitals: 1987-1991. J Surg Oncol 1997; 64:
242-245.
2. Fong
Y, Blumgart LH, Lin E, et al. Outcome of treatment for distal bile duct cancer.
Br J Surg 1996; 83: 1712-1715.
3. Krokidis
M, Hatzidakis A. Percutaneous minimally invasive treatment of malignant
biliary strictures: Current status. Cardiovasc Intervent Radiol 2014;
37(2): 316-323.
4. Assimakopoulos
SF, Scopa CD, Vagianos CE. Pathophysiology of increased intestinal
permeability in obstructive jaundice. World J Gastroenterol 2007; 13:
6458-6464.
5. Walter
T, Ho CS, Horgan AM, et al. Endoscopic or percutaneous biliary drainage for
Klatskin tumors? J Vasc Interv Radiol 2013; 24(1): 113-121.
6. Kloek
JJ, van der Gaag NA, Aziz Y, et al. Endoscopic and percutaneous preoperative
biliary drainage in patients with suspected hilar cholangiocarcinoma. J
Gastrointest Surg 2010; 14(1): 119-125.
7. Pancreatic
Section, British Society of Gastroenterology, Pancreatic Society of Great
Britain and Ireland, Association of Upper Gastrointestinal Surgeons of Great
Britain and Ireland, Royal College of Pathologists, Special Interest Group for
Gastro-intestinal Radiology. Guidelines for the management of patients with pancreatic
cancer periampullary and ampullary carcinomas. Gut 2005; 54 (suppl 5):
v1–v16.
8. Lacomis
JM, Baron RL, Oliver JH, et al. Cholangiocarcinoma: Delayed CT contrast
enhancement patterns. Radiology 1997; 203: 98-104.
9. Guibaud
L, Bret PM, Reinhold C, et al. Bile duct obstruction and choledocholithiasis:
Diagnosis with MR cholangiography. Radiology 1995; 197: 109- 115.
10. Bismuth H, Corlette
MB. Intrahepatic cholangioenteric anastomosis in carcinoma of the hilus of the
liver. Surg Gynecol Obstet 1975; 140: 170-178.
11. Okuda K, Tanikawa K,
Emura T, et al. Nonsurgical, percutaneous transhepatic
cholangiography-diagnostic significance in medical problems of the liver. Am
J Dig Dis 1974; 19(1): 21-36.
12. Bismuth H, Castaing
D, Traynor O. Resection or palliation: Priority of surgery in the treatment of
hilar cancer. World J Surg 1988; 12(1): 39-47.
13. Nilsson U, Evander
A, Ihse I, et al. Percutaneous transhepatic cholangiography and drainage. Risks
and complications. Acta Radiol Diagn (Stockh) 1983; 24(6): 433-439.
14. Hatzidakis A, Adam
A. The interventional radiological management of cholangio-carcinoma. Clinical
Radiology 2003; 58: 91-96.
15. Fritscher-Ravens A,
Broering DC, Sriram PVJ, et al. EUSguided fine-needle cytodiagnosis of hilar
cholangiocarcinoma: A case series. Gastrointest Endosc 2000; 52:
534-540.
16. De Bellis M, Sherman
S, Fogel EL, et al. Tissue sampling at ERCP in suspected malignant biliary
strictures (part 2). Gastrointest Endosc 2002; 56: 720-730.
17. Park JG, Jung GS,
Yun JH, et al. Percutaneous transluminal forceps biopsy in patients suspected
of having malignant biliary obstruction: factors influencing the outcomes of
271 patients. Eur Radiol 2017; 27. doi: 10.1007/s00330-017-4796-x. [Epub
ahead of print]
18. Adam A. Metallic
biliary endoprostheses. Cardiovasc Intervent Radiol 1994; 17: 127-132.
19. Rossi P, Bezzi M,
Rossi M, et al. Metallic stents in malignant biliary obstruction: Results of a
Multicenter European Study of 240 patients. J Vasc Interv Radiol 1994;
5: 279-285.
20. Hatzidakis A, Tsetis
D, Chrysou E, et al. Nitinol stents for palliative treatment of malignant
obstructive jaundice. Should we stent the sphincter of Oddi in every case? Cardiovasc
Intervent Radiol 2001; 24: 245-248.
21. Lammer J, Hausegger
KA, Flückiger F, et al. Common bile duct obstruction due to malignancy:
Treatment with plastic versus metal stents. Radiology 1996;
201(1): 167-172.
22. Brountzos EN,
Ptochis N, Panagiotou I, et al. A survival analysis of patients with malignant
biliary strictures treated by percutaneous metallic stenting. Cardiovasc
Intervent Radiol 2007; 30:66-73.
23. Kaskarelis IS,
Papadaki MG, Papageorgiou GN, et al. Long-term follow-up in patients with
malignant biliary obstruction after percutaneous placement of uncovered
wallstent endoprostheses. Acta Radiol 1999; 40: 528-533.
24. Hatzidakis A,
Krokidis M, Kalbakis K, et al. ePTFE/ FEP-covered metallic stents for palliation
of malignant biliary disease: can tumor ingrowth be prevented? Cardiovasc
Intervent Radiol 2007; 30(5): 950-958.
25. Hausegger KA,
Kleinert R, Lammer J, et al. Biliary obstruction: Histologic findings after
treatment with self-expandable stents. Radiology 1992; 185: 461-464.
26. Boguth L, Tatalovic
S, Antonucci F, et al. Malignant biliary obstruction: Clinical and
histopathologic correlation after treatment with self-expanding metal
prostheses. Radiology 1994; 192: 669-674.
27. Krokidis M,
Hatzidakis A. ePTFE/FEP covered metal stents in malignant biliary disease. In:
Fanelli F (ed). Biliary Disease and Advanced Therapies using ePTFE/ FEP Covered
Stents. Minerva Medica Turin 2014: 23-38.
28. Saito H,
Sakurai Y, Takamura A, et al. Biliary endoprosthesis using Gore Tex covered
expandable metallic stents: Preliminary clinical evaluation. [Article in Japanese].
Nihon Igaku Hoshasen Gakkai Zasshi 1994; 54(2): 180-182.
29. Thurnher SA, Lammer
J, Thurnher MM, et al. Covered self-expanding transhepatic biliary stents:
Clinical pilot study. Cardiovasc Intervent Radiol 1996; 19: 10-14.
30. Rossi P, Bezzi M,
Salvatori FM, et al. Clinical experience with covered Wallstents for biliary
malignancies: 23-month follow-up. Cardiovasc Intervent Radiol 1997; 20:
441-447.
31. Hausegger KA,
Thurnher S, Bodendorfer G, et al. Treatment of malignant biliary obstruction
with polyurethane covered Wallstents. AJR Am J Roentgenol 1998; 170(2):
403-408.
32. Kanasaki S, Furukawa
A, Kane T, et al. Polyurethane-covered nitinol Strecker stents as primary
palliative treatment of malignant biliary obstruction. Cardiovasc Intervent
Radiol 2000; 23: 114-120.
33. Miyayama S, Matsui
O, Terayama T, et al. Covered Gianturco stents for malignant biliary
obstruction: Preliminary clinical evaluation. J Vasc Interv Radiol 1997;
8: 641-648.
34. Han YM, Jin GY, Lee
S, et al. Flared Polyurethane-covered Self expandable Nitinol Stent for
Malignant Biliary Obstruction. J Vasc Interv Radiol 2003; 14:
1291-1301.
35. Isayama H, Komatsu
Y, Tsujino T, et al. Polyurethane-covered metal stent for management of distal
malignant biliary obstruction. Gastrointest Endosc 2002; 55: 366-370.
36. Isayama H, Komatsu
Y, Tsujino T, et al. A prospective randomized study of “covered” vs. “uncovered”
diamond stents for the management of distal malignant biliary obstruction. Gut
2004; 53: 729-734.
37. Krokidis M, Fanelli
F, Orgera G, et al. Percutaneous palliation of pancreatic head cancer:
Randomized comparison of ePTFE/FEP-covered vs. uncovered nitinol
biliary stents. Cardiovasc Intervent Radiol 2011; 34(2): 352-361.
38. Krokidis M, Fanelli
F, Orgera G, et al. Percutaneous treatment of malignant jaundice due to
extrahepatic cholangiocarcinoma: Covered Viabil stent vs. uncovered
Wallstents. Cardiovasc Intervent Radiol 2010; 33: 97-106.
39. Krokidis M, Orgera
G, Fanelli F, et al. Covered biliary metal stents: Which, where, when? Gastrointest
Endosc 2011; 74(5): 173-1174.
40. Adam A, Chetty N,
Roddie M, et al. Self-expandable stainless steel endoprostheses for treatment
of malignant bile duct obstruction. AJR Am J Roentgenol 1991; 156(2):
321-325.
41. Hatzidakis AA,
Charonitakis E, Athanasiou A, et al. Sedation and analgesia in patients
undergoing percutaneous transhepatic biliary drainage. Clin Radiol 2003;
58(2): 121-127.
42. Inal M, Akgul E,
Seydaoglu G. Percutaneous placement of biliary metallic stents in patients with
malignant hilar obstruction: Unilobar vs. bilobar drainage. J Vasc
Interv Radiol 2003;14: 1409-1416.
43. Li M, Wu W, Yin Z,
et al. Unilateral versus bilateral biliary drainage for malignant hilar
obstruction: A systematic review and meta-analysis. [Article in Chinese] Zhonghua
Gan Zang Bing Za Zhi 2015; 23(2): 118-123.
44. De Palma GD, Galloro
G, Siciliano S, et al. Unilateral versus bilateral endoscopic hepatic duct
drainage in patients with malignant hilar biliary obstruction: Results of a
prospective, randomized, and controlled study. Gastrointest Endosc 2001;
53(6): 547-553.
45. Gamanagatti S, Singh
T, Sharma R et al. Unilobar Versus Bilobar Biliary Drainage: Effect on Quality
of Life and Bilirubin Level Reduction. Indian J Palliat Care 2016;
22(1): 50-62.
46. Moss AC, Morris E,
Leyden J, et al. Do the benefits of metal stents justify the costs? A
systematic review and meta-analysis of trials comparing endoscopic stents for
malignant biliary obstruction. Eur J Gastroenterol Hepatol 2007;
19(12): 1119-1124.
47. Hong WD, Chen XW, Wu
WZ, et al. Metal vs. plastic stents for malignant biliary obstruction:
An update meta-analysis. Clin Res Hepatol Gastroenterol 2013; 37(5):
496-500.
48. Al Mahjoub A,
Menahem B, Fohlen A, et al. Preoperative Biliary Drainage in Patients with
Resectable Perihilar Cholangiocarcinoma: Is Percutaneous Transhepatic Biliary
Drainage Safer and More Effective than Endoscopic Biliary Drainage? A
Meta-Analysis. J Vasc Interv Radiol 2017; 28(4): 576-582.
49. Mohkam K, Malik Y,
Derosas C, et al. Percutaneous transhepatic cholangiographic endobiliary
forceps biopsy versus endoscopic ultrasound fine needle aspiration for
proximal biliary strictures: A single-centre experience. HPB (Oxford) 2017;
Mar 13. pii: S1365- 182X(17)30066-7. doi: 10.1016/j.hpb.2017.02.001. [Epub
ahead of print]
50. Huang SY, Philip A,
Richter MD, et al. Prevention and management of infectious complications of
percutaneous interventions. Semin Intervent Radiol 2015; 32(2): 78-88.
51. Venkatesan AM, Kundu
S, Sacks D, et al. Practice guidelines for adult antibiotic prophylaxis during
vascular and interventional radiology procedures. Written by the Standards of
Practice Committee for the Society of Interventional Radiology and Endorsed by
the Cardiovascular Interventional Radiological Society of Europe and Canadian
Interventional Radiology Association. J Vasc Interv Radiol 2010; 21:
1611-1630.
52. Brody LA, Brown KT,
Getrajdman GI, et al. Clinical factors associated with positive bile cultures
during primary percutaneous biliary drainage. J Vasc Interv Radiol 1998; 9: 572-578.
53. Winick AB, Waybill
PN, Venbrux AC. Complications of percutaneous transhepatic biliary interventions.
Tech Vasc Interv Radiol 2001;4: 200-206.
54. Yarmohammadi H and
Covey AM. Percutaneous biliary interventions and complications in malignant
bile duct obstruction. Chin Clin Oncol 2016; 5(5): 68-78.
55. L’Hermine C, Ernst
O, Delemazure O, et al. Arterial complications of percutaneous transhepatic
biliary drainage. Cardiovasc Intervent Radiol 1996; 19(3): 160-164.
56. Rivera-Sanfeliz GM,
Assar OS, LaBerge JM, et al. Incidence of important hemobilia following transhepatic
biliary drainage: Left-sided vs. right-sided approaches. Cardiovasc
Intervent Radiol 2004; 27(2): 137-139.
57. Choi SH, Gwon DI, Ko
GY, et al. Hepatic Arterial Injuries in 3110 Patients Following Percutaneous
Transhepatic Biliary Drainage. Radiology 2011; 261(3): 969-975.
58. Marynissen T, Maleux
G, Heye S, et al. Transcatheter arterial embolization for iatrogenic hemobilia
is a safe and effective procedure: Case series and review of the literature. Eur
J Gastroenterol Hepatol 2012; 24: 905-909.
59. Tsai CC, Chiu KC, Mo
LR, et al. Transcatheter arterial coil embolization of iatrogenic
pseudoaneurysms after hepatobiliary and pancreatic interventions. Hepatogastroenterology
2007; 54(73): 41-46.
60. Dale AP, Khan R,
Mathew A, et al. Hepatic tract embolization after biliary stenting. Is it
worthwhile? Cardiovasc Intervent Radiol 2015; 38(5): 1244-1251.
61. Uller W, Mueller-Wille
R, Grothues D, et al. Gelfoam for closure of large percutaneous transhepatic
and transsplenic puncture tracts in pediatric patients. Rofo 2014;
186(7): 693-697.
62. Park SY, Kim J, Kim
BW, et al. Embolization of percutaneous transhepatic portal venous access
tract with N-butyl cyanoacrylate. Br J Radiol 2014; 87: 20140347.
Ready - Made Citation
Hatzidakis
A, Krokidis M. Percutaneous transhepatic image-guided biliary interventions for
malignant biliary obstructions: Current status. Hell J
Radiol 2017; 2(2): 37-50.